Presentation by Dr. Garland
Dr. Eva Garland began with a data-driven overview of FY25 non-dilutive funding, emphasizing that despite uneven cadence and delays, the award pipeline remains resilient and funding levels are equivalent to prior years. She then translated these trends into actionable insights, describing how the shutdown affects applications already under review, which activities may proceed on active awards, status of the SBIR/STTR program, what solicitations remain active amid reduced agency support (including ARPA-H and BARDA), and the preparation steps that will position applicants for timely submission when operations resume.
FY25 Funding Dynamics. Under the new administration, the granting of new awards has been slow, but the opportunity remains strong. New awards lagged from February to June, then caught up to FY24 by year-end. As Figure 1 shows, the NIH alone issued >$2B more in non-dilutive funding each month in August and September 2025 compared to 2024, indicating that agencies like the NIH are anticipated to release funds quickly once the FY26 budget is in place. Several other agencies reported higher late-year paylines, and most of the feared headwinds never materialized: SBIR/STTR overhead stayed intact and few awards were rescinded. However, there were some significant changes that affected applications, including increased scrutiny of foreign involvement where some proposals were flagged or denied at the Foreign Risk Assessment stage despite strong scientific scores, highlighting the importance of complete and transparent disclosures. Overall, the funding environment remains favorable for applicants who stay engaged and submit well-prepared proposals.
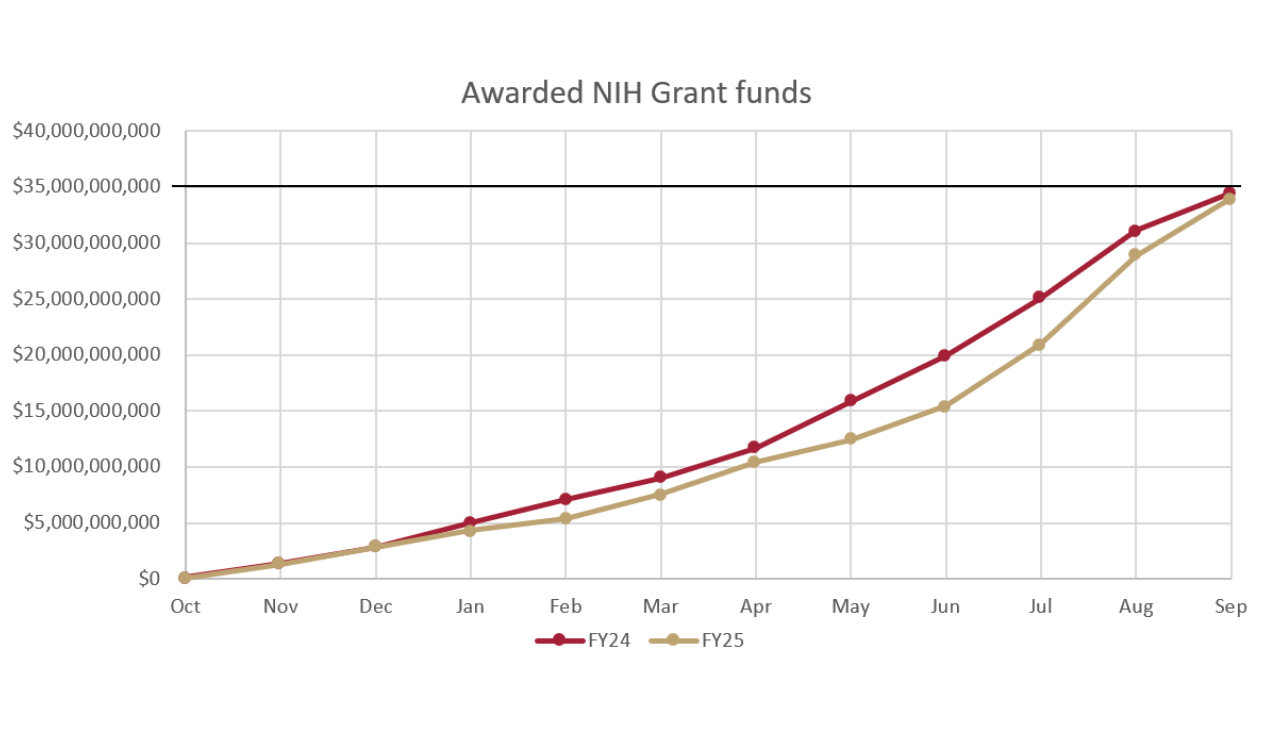
Figure 1
Figure 1 | Awarded NIH Grant Funds. Monthly FY25 NIH awards tracked closely with FY24 through January, then lagged from February before a late-year surge closed the gap, ending near FY24’s ~$35B total.
Status of Applications and Pending Awards. During the federal shutdown, electronic submission systems and help-desk support systems (including SAM.gov, ASSIST/eRA Commons, Research.gov, DSIP, and grants.gov) remain operational but agency staff availability is restricted. Applicants are encouraged to proactively file help desk tickets to time-stamp issues, document communications, and preserve their place in the queue once staff return to duty. While NIH has asked assigned reviewers to continue evaluations to minimize restart delays, Study Sections and Councils will not meet until operations resume, potentially delaying Council decisions and award issuance. For animal work, local IACUC review can proceed, but OLAW approvals are paused and awards contingent on OLAW must wait. Payment systems (e.g., NIH Payment Management System) remain available for drawdowns with short delays, though new Notices of Award generally will not be issued until the government reopens.
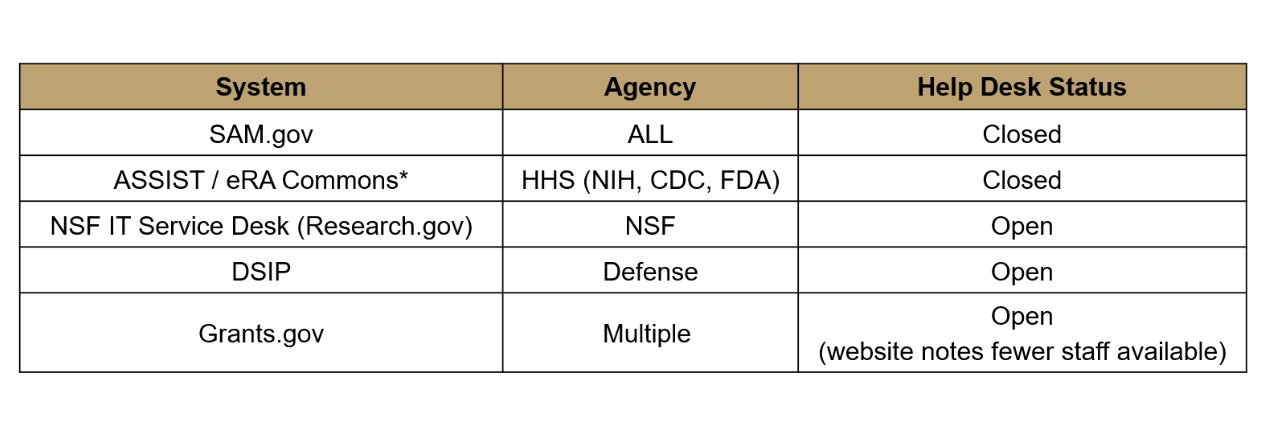
Figure 2
Figure 2 | Status of Online Help Desk Support.
Status of Ongoing Awards. Work on active awards may continue provided no pending agency approval or restriction applies. During the shutdown, PIs should email their Program Officers to time-stamp any requests for scope or budget changes (See Figure 3). The NIH Payment Management System is processing drawdowns, albeit with short delays. Reporting deadlines—including RPPRs, final reports, and tax filings—still apply. While companies are waiting for systems to reopen, this is a good time to ensure their infrastructure is in-place, including:
- Payroll
- Set up vendors and contractors
- Policies & Procedures Manual
- Establish Timekeeping
- Project Cost Accounting System
- Tax prep

Figure 3
Figure 3 | When to Contact a Program Official. Email correspondence with your Program Official during the shutdown is encouraged to document your actions and potentially expedite communication upon reopening.
Status of the SBIR/STTR Program. The Omnibus SBIR/STTR program expired on September 30, 2025. funding for current awards remains, but there will be no new awards issued until the reauthorization. The reauthorization timeline is difficult to predict. Congress discussed a 1 year, or even a 1 month extension before the shutdown, but the measure did not pass the Senate. The future of the program is promising, as SBIR/STTR has STRONG bi-partisan support, and has been in existence since 1982.
Status of Open Solicitations. Companies are encouraged to apply to open solicitations, given that there is a high likelihood of reauthorization. Also, there are likely to be fewer applications, resulting in a higher chance of success.
Current SBIR/STTR solicitations include:
- NSF – last deadline for current solicitation is November 5, 2025
- NIH – various special topics, including those for NIAID, NINDS, NEI, NCATS – next due date is December 9, 2025
- DoD – SBIR BAA 25.4/STTR BAA 25.D Release 12
- pre-release started September 3rd and is currently extended
- topics were to be opened on September 24th, but will now open first Wednesday following program reauthorization
- closing date will be set at a 4-week period after reopening – TIGHT TIMELINE!
With the shutdown, there will be less support available from agencies, so you will need to prepare your application independently. You can take advantage of existing resources, such as EGC’s Winning SBIR/STTR Grants book series (Figure 4).

Figure 4
Figure 4 | EGC’s SBIR/STTR Grant Preparation Resources. Our Winning SBIR/STTR Grants series provides step-by-step guidance, timelines, and checklists to help you plan, budget, and submit competitive proposals independently. All three titles are available for purchase on our website: https://www.evagarland.com/resources/sbir-sttr/.
Tips for Preparing for the Reopening
Tip #1 DOCUMENT any issues that you would normally communicate to Program Staff
During the shutdown, document and time-stamp all issues and every attempt to contact program staff.
Tip #2 ENSURE your grant compliance and audit readiness
For active awards, strengthen post-award compliance by updating FCOI and foreign disclosures, refreshing policies and procedures, verifying project cost accounting and indirect rates, and ensuring GAAP financials and subcontractor monitoring are current.
Tip #3 DRAFT additional grant proposals
Prepare for short turnarounds at the reopening by lining up collaborators, finalizing technical content, and preparing budgets that require input from multipe sources.
Tip #4 Keep ABREAST of Developments in the Federal Landscape
Keep in mind that agencies with approved multi-year budgets may continue to release solicitations, during or shortly after the shutdown ends. These include ARPA-H and BARDA.
Tip #5 Consider ADDITIONAL Sources of Funding
While the federal government is shutdown, look into additional sources of funding for your science – such as through state or local programs such as California Institute for Regenerative Medicine (CIRM) – or from Private Foundations, such as the Gates Foundation, Wellcome Trust, American Heart Association, and the American Cancer Society.
Tip #6 Focus on your SCIENCE – What CAN you do to move it forward during the shutdown?
Lay the foundation for your future success through activities such as generating Preliminary Data, conducting literature reviews, and taking advantage of networking opportunities.
Question and Answer Session
A: Yes! Active solicitations are accepting applications, so you are encouraged to submit to the open deadlines. It is a good idea to submit in advance of the due date due to limited help-desk support. Remember to file a ticket to time-stamp any issues during submission and follow up via email.
A: Study sections aren’t meeting, but Scientific Review Groups were asked to proceed with their review to ensure panels and Council can meet upon re-opening. Hold out hope for proposals that are beyond Council until a formal notice is received; follow up with your PO once operations resume.
A: You can continue working on active awards during the shutdown if there are no pending approvals or restrictions. NIH PMS is operational with short delays. Agency invoice-review payments may move slowly with limited support for troubleshooting. Be sure to include budget-category detail in drawdowns to avoid delays. Document any scope/budget changes now via time-stamped email to your PO.
A: First NCE requests are open; second NCEs require program review and may be delayed. Submit and notify your Program Officer via email ASAP.
A: Yes, processing delays are expected due to limited staff but the portal is open for new registrations. For ongoing registrations, submit a help-desk ticket now to enter the queue and document communications to retain proof of timely submission.
A: Restrictions and delays are commonly tied to human subjects research (IRB approval, training certificates), animal research (IACUC and OLAW approval), and accounting system compliance. Assess and prepare documentation to address these restrictions.
A: Inventory every party involved in the project (investors, vendors, subawardees, consultants, and especially key personnel) and map where work, funds, data, and IP access will occur. Treat risk as country-agnostic and flag any non-U.S. ties or non-U.S. performance. Given limited up-front rules and reported denials for foreign risk, take a conservative approach: keep work stateside when possible, adjust roles if needed (e.g., avoid listing higher-risk individuals as key personnel), and fully disclose all foreign affiliations/components with dated documentation.
A: Yes! You can submit a Project Pitch now, but responses may be delayed pending Program Manager review.
